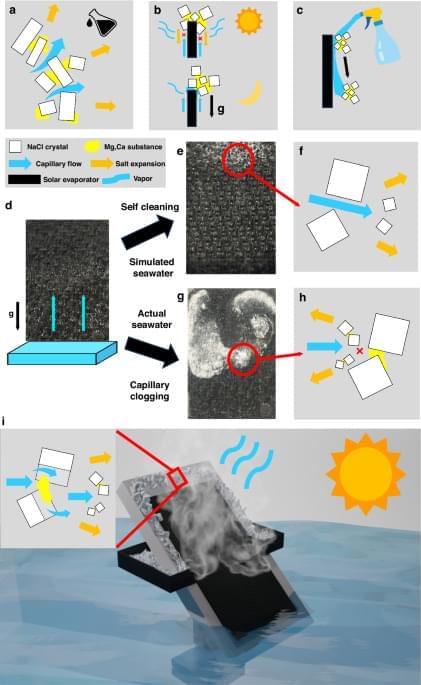
This study examined the potential of narrow-bandgap (Perovskite-based tandem solar cells are a promising photovoltaic (PV) technology to exceed the Shockley–Queisser limit of single-junction solar cells. Perovskite/Si tandem solar cells have been intensively studied, demonstrating a record power conversion efficiency (PCE) of 34.6% [1]. In contrast, the certified record PCE of perovskite/Cu(In, Ga)Se2 (CIGS) tandem solar cells remains 24.6% with a reported efficiency of 24.9% [1, 2]. Theoretical calculations for double-junction tandem solar cells using a detailed balance model indicate that the bandgap (Eg) combinations of 1.12 eV (for a bottom cell) and 1.70 eV (for a top cell) or 0.90 to 1.04 eV (for a bottom cell) and 1.58 to 1.67 eV (for a top cell) can yield a maximum theoretical tandem efficiency [3, 4]. Wide-bandgap perovskite (with Eg equal to or greater than 1.7 eV) has been actively studied for tandem application with Si (Eg = 1.12 eV), the most successful solar cell technology to date as a bottom cell. However, previous studies have shown that wide-bandgap perovskite suffers from substantial open-circuit voltage (VOC) loss due to halide segregation [5], and the maximum PCEs of single-junction perovskite cells have been produced by perovskite with Eg between 1.52 and 1.63 eV [6– 8]. The bandgap of CIGS can be tuned between 1.01 and 1.68 eV by adjusting the Ga/(Ga+In) (GGI) ratio and through tuning of bandgap grading profile [9]. Employing a narrow-bandgap CIGS close to 1.00 eV as a bottom cell is advantageous to use the most efficient, conventional bandgap perovskite as the top cell. Therefore, unlike Si, the bandgap tunability of CIGS offers an opportunity for perovskite/CIGS to attain a greater ultimate performance than perovskite/Si tandem solar cells. Han et al. [10] introduced a thick indium-doped tin oxide (ITO) recombination layer to bury the intrinsic surface roughness of CIGS, followed by chemical mechanical polishing to prepare a smooth surface for the subsequent solution process of perovskite, attaining a certified PCE of 22.4%. Albrecht and coworkers have improved the PCE of perovskite/CIGS tandem solar cells by modifying the hole transport layer (HTL). In their earlier work, a NiOx/PTAA bilayer was utilized to form a uniform HTL on CIGS bottom cells. Recently, a self-assembled monolayer such as 2PACz and Me-4PACz was used, which can enhance the device performance of single-junction perovskite solar cell and its perovskite/CIGS tandem counterpart, achieving a certified PCE of 24.2% [2, 11 – 13].
Most recent studies on perovskite/CIGS tandem solar cells have focused on optimizing the perovskite top cell. In contrast, all CIGS bottom cells include an absorber with a double-graded (DG) bandgap profile optimized around the bandgap of ~1.1 eV. The DG bandgap profile has been adapted primarily for CIGS absorbers prepared by thermal evaporation, which has resulted in high-performing CIGS solar cells with PCEs up to 23.4% [14], and it has proven to be an effective strategy for enhancing performance, optimized for “single-junction” CIGS; however, it has not been determined whether DG would be the ideal configuration for tandem applications. Kim et al. [15] used single-graded (SG) CIGS with a bandgap close to 1.0 eV, where the band grading is only formed on the backside of the absorber. They employed dual alkali post-deposition treatment (PDT) with KF and CsF, demonstrating a CIGS solar cell with a PCE of 20.