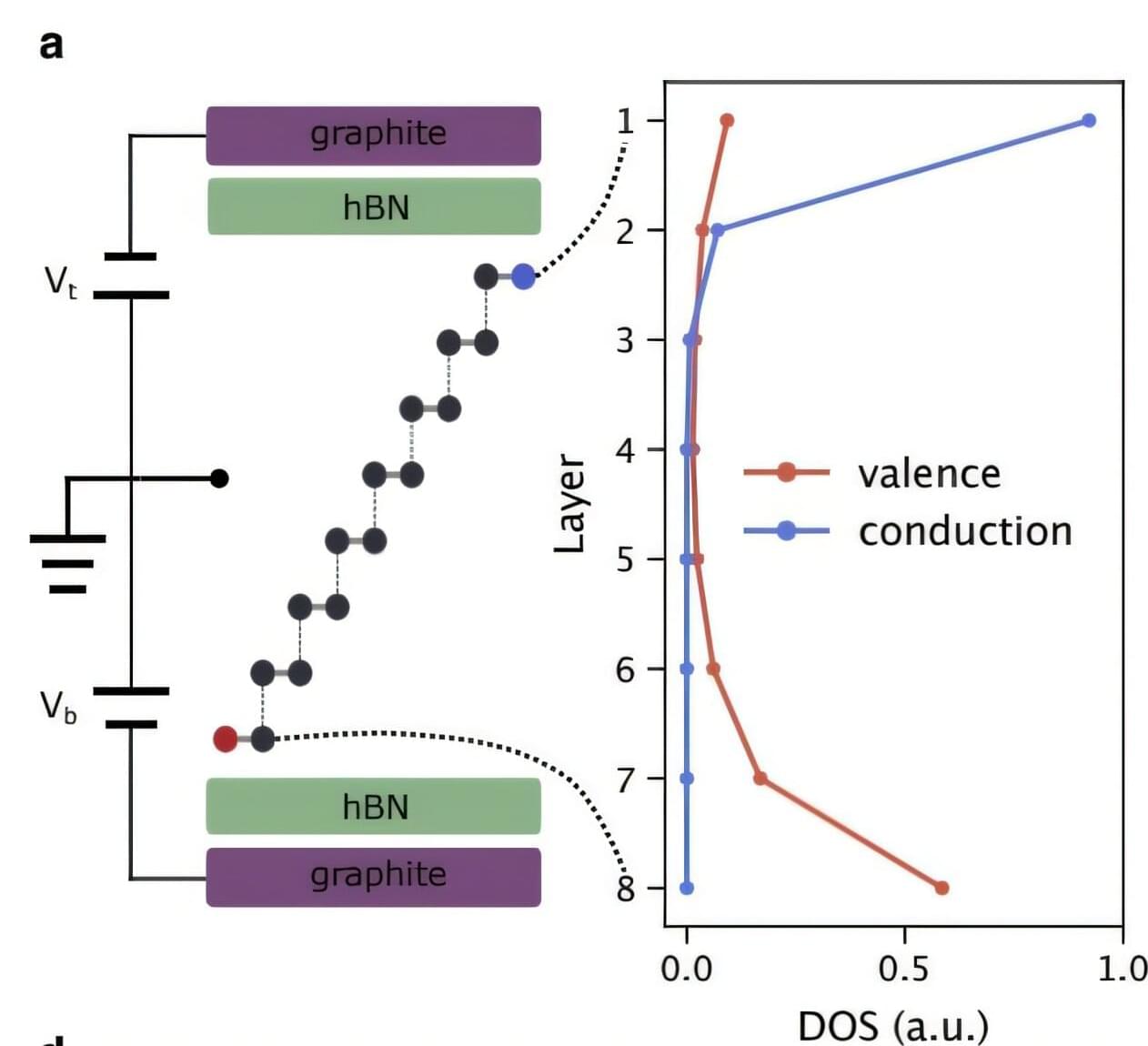
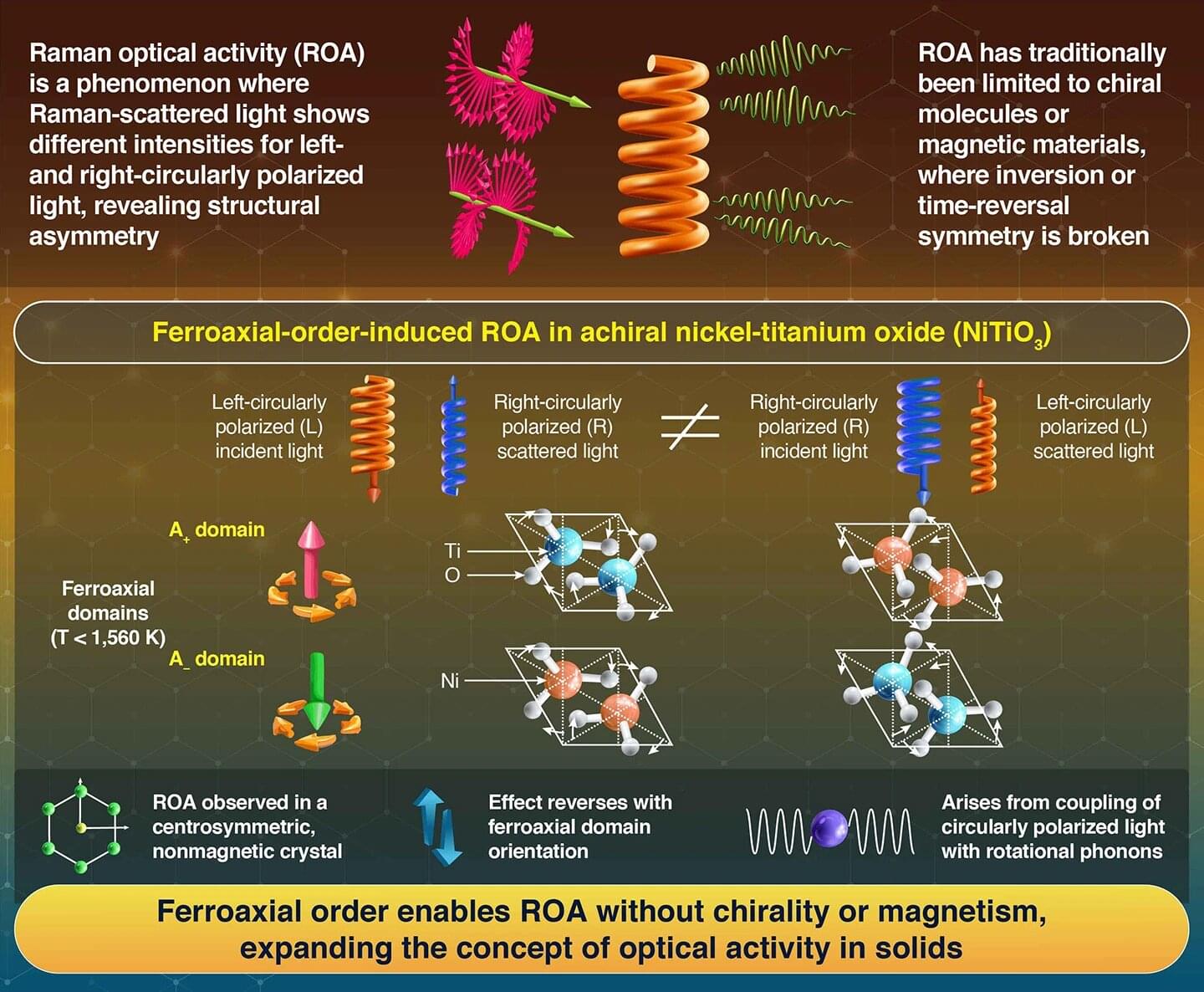
Experimental attosecond science is built around the ability to generate and control light flashes lasting billionths of a billionth of a second. Such extreme pulses can be created through high harmonic generation (HHG), where an intense laser field drives electrons out of atoms or solids and then forces them back, releasing bursts of extreme ultraviolet radiation. Techniques like this have transformed our ability to observe electron motion on its natural timescale.
To extract information from such ultrafast processes, physicists often rely on attosecond interferometry. By combining a strong laser field with a weaker second colour, different electron trajectories are made to interfere, imprinting timing and phase information onto the emitted harmonics. Over recent years, these schemes have become standard tools for attosecond metrology and spectroscopy.