Category: biotech/medical – Page 1,542
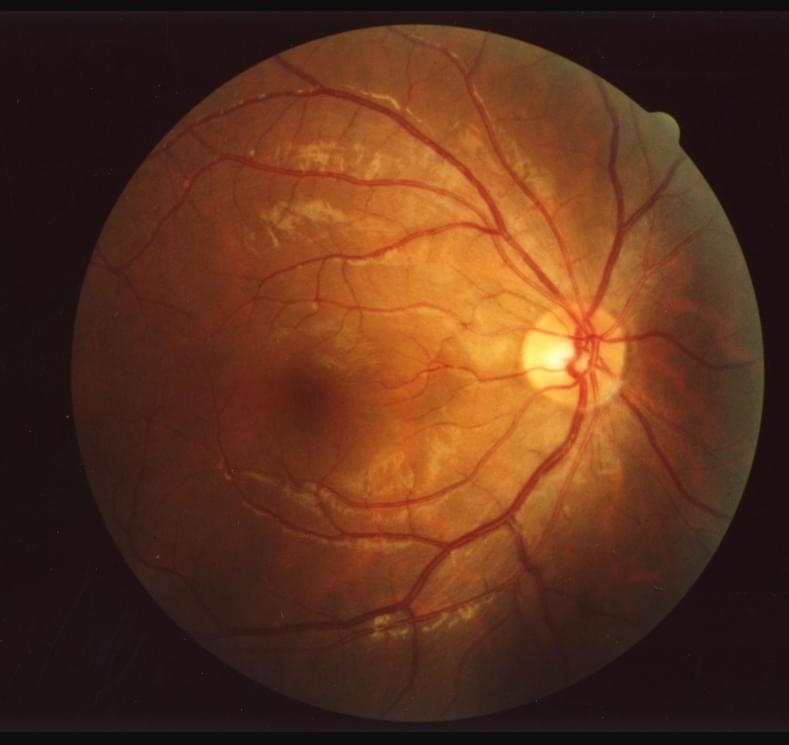
Deep-learning aging clock tracks human aging, detects eye and other diseases from retinal images
A team of biomedical researchers has developed a non-invasive, more accurate, and inexpensive “aging clock” for tracking and slowing human aging by examining retinal images and using trained deep-learning models of the eye’s fundus (the deepest area of the eye), using a new “eyeAge” system.
The researchers are affiliated with Buck Institute for Research on Aging, Google Research, Google Health, Zuckerberg San Francisco General Hospital, Post Graduate Institute of Medical Education, and Research (India), and University of California, San Francisco.
Tracking eye changes that accompany aging and age-related diseases: the eyeAge system.
Scientists Find Antibiotic-Free Way to Treat Drug-Resistant Infections
Scientists have found an antibiotic-free way of treating ‘golden staph’ skin infections that are the scourge of some cancer patients, and a threat to hospital-goers everywhere.
The lab study from researchers at the University of Copenhagen utilized an artificial version of an enzyme that’s naturally produced by bacteriophages (viruses that infect bacteria), and used it to eradicate Staphylococcus aureus, or golden staph, in biopsy samples from people with skin lymphoma.
“To people who are severely ill with skin lymphoma, staphylococci can be a huge, sometimes insoluble problem, as many are infected with a type of Staphylococcus aureus that is resistant to antibiotics,” explains immunologist Niels Ødum of the University of Copenhagen.
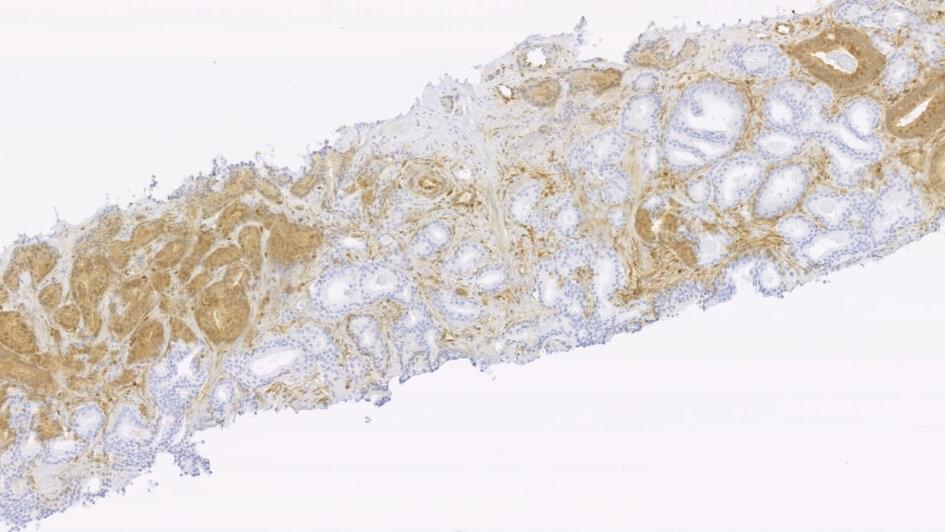
Biological markers identified as powerful predictors of prostate cancer relapse following radiotherapy
Two key proteins linked to cell division can reliably predict disease recurrence in prostate cancer after radiotherapy treatment, according to new research.
Using an inexpensive and widely available technique in the clinic, the researchers evaluated a range of proteins in prostate tumor biopsies and determined that the phosphatase and tensin homolog (PTEN) and geminin proteins are key markers associated with cancer relapse after radiotherapy.
Based on their results, the team at The Institute of Cancer Research, London, reported that patients with tumors showing loss of PTEN were almost three times more likely to experience recurrence than those with ‘normal’ PTEN. Similarly, the data showed a 70 percent increase in the likelihood of experiencing recurrence in patients with tumors that had a 10 percent increase in geminin.
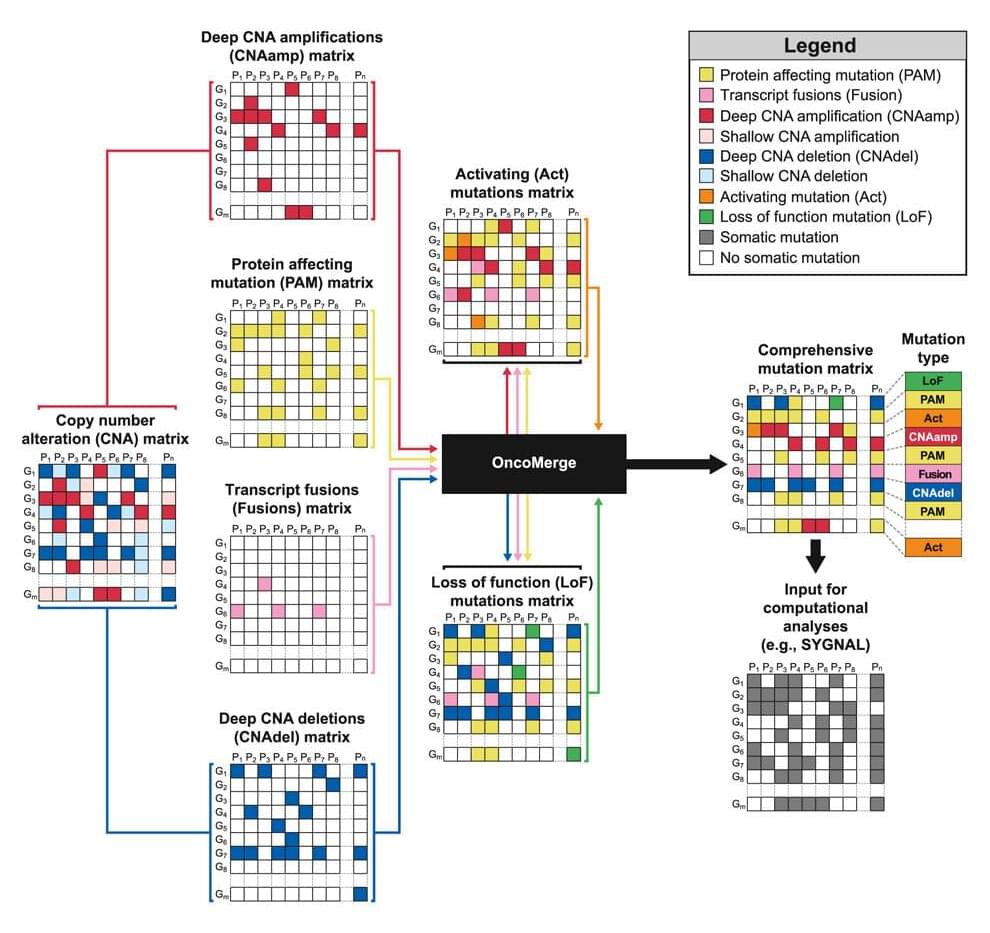
Genetic analysis tool developed to improve cancer modeling
Lifestyle behaviors such as eating well and exercising can be significant factors in one’s overall health. But the risk of developing cancer is predominantly at the whim of an individual’s genetics.
Our bodies are constantly making copies of our genes to produce new cells. However, there are occasional mistakes in those copies, a phenomenon geneticists call mutation. In some cases, these mistakes can alter proteins, fuse genes and change how much a gene gets copied, ultimately impacting a person’s risk of developing cancer. Scientists can better understand the impact of mutations by developing predictive models for tumor activity.
Christopher Plaisier, an assistant professor of biomedical engineering in the Ira A. Fulton Schools of Engineering at Arizona State University, is developing a software tool called OncoMerge that uses genetic data to improve cancer modeling technology.
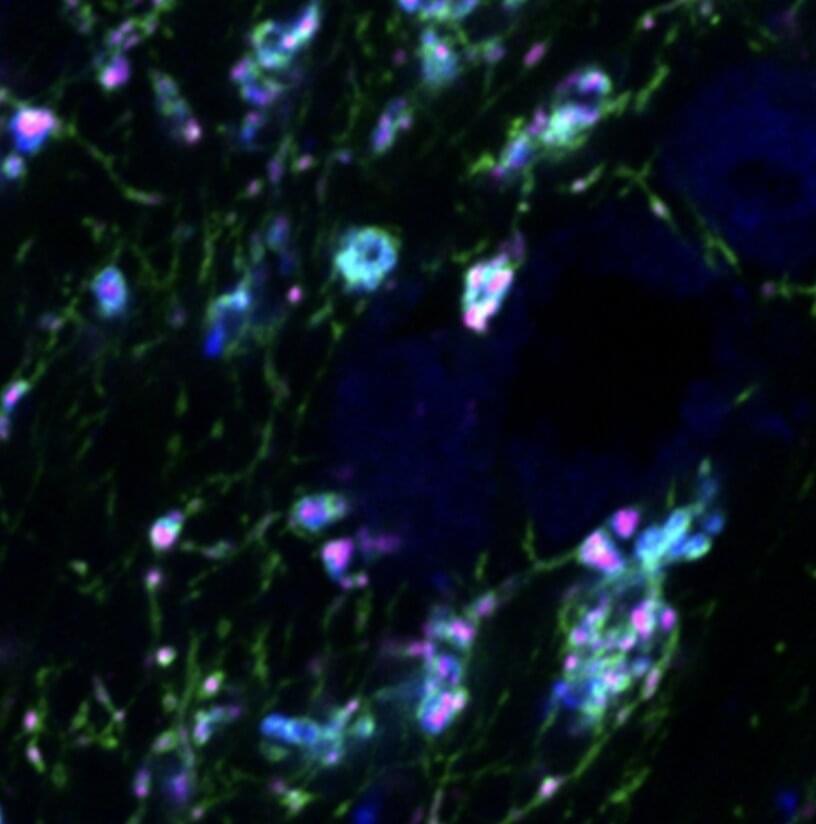
Serotonin gates the transfer of visual information from the eyes to the thalamus
Humans are known to perceive the environment around them differently based on the situation they are in and their own feelings and sensations. Internal states, such as fear, arousal or hunger can thus affect the ways in which sensory information is processed and registered by the brain.
Researchers at Beth Israel Deaconess Medical Center, Boston Children’s Hospital, and Peking University have recently carried out a study investigating the possible effects of serotonin, a neurotransmitter known to regulate sleep, mood, sexual desire, and other inner states, in the processing of visual information. Their findings, published in Neuron, suggest that serotonergic neurons in the brainstem (i.e., the central trunk of the mammalian brain) gate the transfer of visual information from the eyes to the thalamus, an egg-shaped area of the brain.
“Internal states are known to affect sensory perception and processing, but this was generally thought to occur in the cortex or thalamus,” Chinfei Chen, one of the researchers who carried out the study, told Medical Xpress. “One of our previous studies revealed that arousal can suppress certain visual information channels at an earlier stage of the visual pathway–at the connection between the mouse retina and the thalamus, before the information even reaches the brain. This form of ‘filtering’ of information suggests a very efficient means of processing only relevant information.”
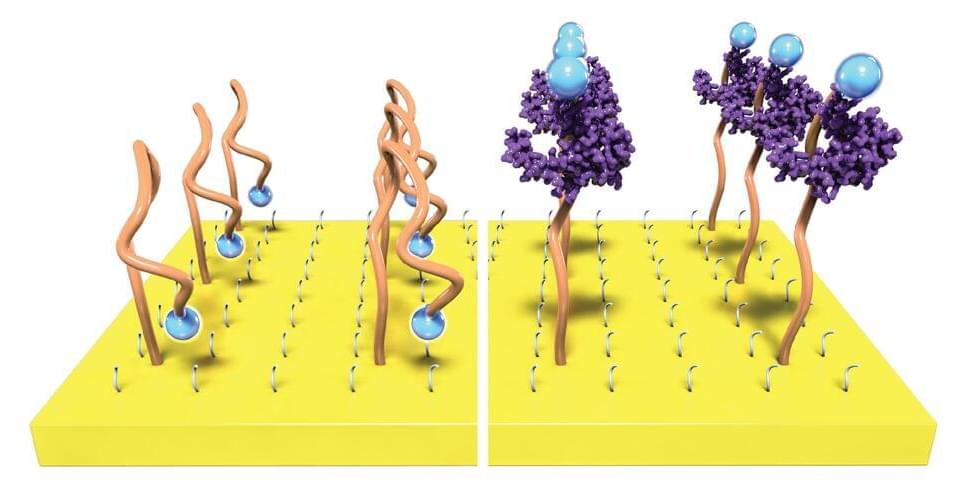
Plastic transistor amplifies biochemical sensing signal
The molecules in our bodies are in constant communication. Some of these molecules provide a biochemical fingerprint that could indicate how a wound is healing, whether or not a cancer treatment is working or that a virus has invaded the body. If we could sense these signals in real time with high sensitivity, then we might be able to recognize health problems faster and even monitor disease as it progresses.
Now Northwestern University researchers have developed a new technology that makes it easier to eavesdrop on our body’s inner conversations.
While the body’s chemical signals are incredibly faint—making them difficult to detect and analyze—the researchers have developed a new method that boosts signals by more than 1,000 times. Transistors, the building block of electronics, can boost weak signals to provide an amplified output. The new approach makes signals easier to detect without complex and bulky electronics.

The quantum revolution: Brain waves
Presented by Madhumita Murgia and John Thornhill, produced by Josh Gabert-Doyon and Edwin Lane. Executive producer is Manuela Saragosa. Sound design by Breen Turner and Samantha Giovinco. Original music by Metaphor Music. The FT’s head of audio is Cheryl Brumley. Special thanks to The Hospital for Sick Children.
We’re keen to hear more from our listeners about this show and want to know what you’d like to hear more of, so we’re running a survey which you can find at ft.com/techtonicsurvey. It takes about 10 minutes to complete and you will be in with a chance to win a pair of Bose QuietComfort earbuds.